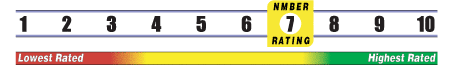
Each capsule contains: Chitosan 800 mg. Other Ingredients: Gelatin.
Brand name products often contain multiple ingredients. To read detailed information about each ingredient, click on the link for the individual ingredient shown above.

In 2004, Canada began regulating natural medicines as a category of products separate from foods or drugs. These products are officially recognized as "Natural Health Products." These products include vitamins, minerals, herbal preparations, homeopathic products, probiotics, fatty acids, amino acids, and other naturally derived supplements.
In order to be marketed in Canada, natural health products must be licensed. In order to be licensed in Canada, manufacturers must submit applications to Health Canada including information about uses, formulation, dosing, safety, and efficacy.
Products can be licensed based on several criteria. Some products are licensed based on historical or traditional uses. For example, if an herbal product has a history of traditional use, then that product may be acceptable for licensure. In this case, no reliable scientific evidence is required for approval.
For products with non-traditional uses, some level of scientific evidence may be required to support claimed uses. However, a high level of evidence is not necessarily required. Acceptable sources of evidence include at least one well-designed, randomized, controlled trial; well-designed, non-randomized trials; cohort and case control studies; or expert opinion reports.
Finished products licensed by Health Canada must be manufactured according to Good Manufacturing Practices (GMPs) as outlined by Health Canada.
Below is general information about the effectiveness of the known ingredients contained in the product Libracol. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
INSUFFICIENT RELIABLE EVIDENCE to RATE
Below is general information about the safety of the known ingredients contained in the product Libracol. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
POSSIBLY SAFE. ..when used orally, short-term. Chitosan has been used with apparent safety in clinical studies at a dose of up to 1.35 grams daily for up to 3 months (1942,9609,9610,10022,10023,10024,10025,11307,13171,14314)(15126,92781,97708). ...when used topically, short-term (1944,1945,4269,4270,97712,106521).
PREGNANCY AND LACTATION:
Insufficient reliable information available; avoid using.
Below is general information about the interactions of the known ingredients contained in the product Libracol. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
Chitosan can reduce the absorption of acyclovir, potentially increasing the risk for treatment failure.
Clinical research in humans shows that taking chitosan along with acyclovir 200 mg reduces acyclovir absorption. Concomitant administration of chitosan 400 mg or 1000 mg reduced the acyclovir area under the curve (AUC) and peak plasma concentration by about 30% and 40%, respectively, compared with control. Concomitant administration with chitosan 1000 mg also increased time to peak concentration from 1 hour to 2 hours (92780). In vitro research suggests that the mechanism for reduced absorption is due to acyclovir entrapment in chitosan-mucus complexes, which reduces intestinal absorption (112352).
|
Theoretically, chitosan might increase the risk of bleeding when taken with warfarin.
In a case report, a patient taking warfarin had a significantly increased international normalized ratio (INR) after starting chitosan 1200 mg daily. The INR normalized after chitosan was discontinued and vitamin K was administered. The patient once again started taking chitosan and again had a significant increase in INR. The INR stabilized again once chitosan was discontinued (15909). Researchers theorize that this interaction might occur because chitosan decreases absorption of fat-soluble vitamins, including vitamin K, which could increase the anticoagulant effect of warfarin.
|
Below is general information about the adverse effects of the known ingredients contained in the product Libracol. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
General
...Orally and topically, chitosan seems to be well tolerated, short-term.
Most Common Adverse Effects:
Orally: Constipation, diarrhea, flatulence, epigastric discomfort, and nausea.
Dermatologic ...In one clinical trial, a subject with kidney failure reported itching during 12 weeks of oral chitosan treatment (1942). It is not clear if this was related to the underlying renal failure or the use of oral chitosan.
Gastrointestinal ...Orally, chitosan has been reported to cause epigastric discomfort, constipation, flatulence, diarrhea, nausea, and dryness of the throat (1942,3243,9986,11307,14314,41688,92781,100170). Excessive discharge of fat in the feces, also known as steatorrhea, has been reported with chitosan therapy (41724,41726). Theoretically, chitosan may alter the normal intestinal flora via antimicrobial activity, which could interfere with lipid digestibility and bile acid metabolism, leading to the growth of resistant pathogens (41687,41709,41725).
Musculoskeletal ...Orally, chitosan has been reported to cause swollen heels and wrists in two patients (41688).
Neurologic/CNS ...Headaches have been reported in patients taking oral chitosan (41688).
Ocular/Otic ...Topically, an eye drop containing chitosan-N-acetylcysteine has been reported to cause itching and irritation of the eyes (97710).
