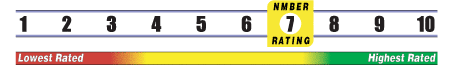
Each 0.25 tsp serving contains: Inositol Niacinate 500 mg.
Brand name products often contain multiple ingredients. To read detailed information about each ingredient, click on the link for the individual ingredient shown above.

In 2004, Canada began regulating natural medicines as a category of products separate from foods or drugs. These products are officially recognized as "Natural Health Products." These products include vitamins, minerals, herbal preparations, homeopathic products, probiotics, fatty acids, amino acids, and other naturally derived supplements.
In order to be marketed in Canada, natural health products must be licensed. In order to be licensed in Canada, manufacturers must submit applications to Health Canada including information about uses, formulation, dosing, safety, and efficacy.
Products can be licensed based on several criteria. Some products are licensed based on historical or traditional uses. For example, if an herbal product has a history of traditional use, then that product may be acceptable for licensure. In this case, no reliable scientific evidence is required for approval.
For products with non-traditional uses, some level of scientific evidence may be required to support claimed uses. However, a high level of evidence is not necessarily required. Acceptable sources of evidence include at least one well-designed, randomized, controlled trial; well-designed, non-randomized trials; cohort and case control studies; or expert opinion reports.
Finished products licensed by Health Canada must be manufactured according to Good Manufacturing Practices (GMPs) as outlined by Health Canada.
Below is general information about the effectiveness of the known ingredients contained in the product Nianate Powder. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
INSUFFICIENT RELIABLE EVIDENCE to RATE
Below is general information about the safety of the known ingredients contained in the product Nianate Powder. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
Below is general information about the interactions of the known ingredients contained in the product Nianate Powder. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
Theoretically, concomitant use of inositol nicotinate with anticoagulant or antiplatelet drugs might increase the risk of bleeding.
|
Theoretically, inositol nicotinate might increase blood glucose levels and may diminish the effects of antidiabetes drugs.
|
Theoretically, concomitant use of inositol nicotinate and statins might increase the risk of myopathy and rhabdomyolysis.
Inositol nicotinate is metabolized to niacin in the body (496). Some case reports have raised concerns that niacin might increase the risk of myopathy and rhabdomyolysis when combined with statins (14508,25918) However, a significantly increased risk of myopathy has not been demonstrated in clinical trials (7388,11689,12033,14509).
|
Theoretically, concomitant use of inositol nicotinate and transdermal nicotine might increase the risk of flushing and dizziness.
|
Below is general information about the adverse effects of the known ingredients contained in the product Nianate Powder. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
General
...Orally, inositol nicotinate is generally well tolerated.
Most Common Adverse Effects:
Orally: Abdominal pain, constipation, diarrhea, flushing, heartburn, liver enzyme elevation, nausea, vomiting.
Cardiovascular ...Orally, inositol nicotinate is converted to niacin and is therefore likely to cause similar side effects as niacin (496). A prostaglandin-mediated flushing reaction is a common adverse effect of oral niacin. A large pooled analysis of clinical studies shows that up to 70% of people may experience flushing with oral niacin (96211). Although flushing can occur with doses of niacin as low as 30 mg daily, it is more common with the larger doses of 500-2000 mg used for treatment of dyslipidemia (4889,26089,93341,93345). Inositol nicotinate is often promoted as a "no-flush" niacin; however, this benefit has not been demonstrated in clinical trials.
Dermatologic ...Orally, inositol nicotinate is converted to niacin and is therefore likely to cause similar side effects as niacin, including pruritus (496).
Endocrine ...Orally, inositol nicotinate is converted to niacin and is therefore likely to cause similar side effects as niacin (496). Niacin can impair glucose tolerance in a dose-dependent manner. Dosages of niacin 3-5 grams daily appear to increase blood glucose in patients with or without diabetes, while dosages of 1.5 grams or less have minimal effects (12033). Because inositol nicotinate is converted to niacin, it may also impair glucose tolerance (496).
Gastrointestinal ...Orally, inositol nicotinate is converted to niacin and is therefore likely to cause similar side effects as niacin, including gastrointestinal complaints such as nausea, vomiting, bloating, heartburn, diarrhea, and constipation (496). These adverse effects from niacin may be reduced by taking it with meals or antacids, and usually disappear within two weeks of continued therapy (4851,11690,26094).
Hematologic ...Orally, inositol nicotinate is converted to niacin and is therefore likely to cause similar side effects as niacin, including hyperuricemia (496).
Hepatic ...Orally, inositol nicotinate is converted to niacin and is therefore likely to cause similar side effects as niacin (496). Niacin is associated with elevated liver function tests and jaundice, especially with doses of 3 grams daily or more, and when doses are rapidly increased (4458,4863,6243,11690). Because inositol nicotinate is converted to niacin, it may also cause hepatotoxicity (496)
