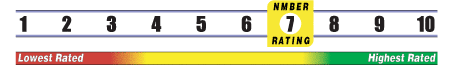
| Ingredients | Amount Per Serving |
|---|---|
| 250 mg | |
|
Proprietary Blend
(Proprietary Blend Note: 5 billion CFU )
|
|
|
(B. lactis )
|
|
|
(L. acidophilus )
|
|
|
(B. longum )
|
|
|
(L. lactis )
|
|
|
(L. rhamnosus )
|
Microcrystalline Cellulose (Form: Plant Fiber), Hydroxypropyl Methylcellulose Note: vegetable capsule, Magnesium Stearate, Silica
Below is general information about the effectiveness of the known ingredients contained in the product Probiotic with Digestive Enzymes. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
INSUFFICIENT RELIABLE EVIDENCE to RATE
INSUFFICIENT RELIABLE EVIDENCE to RATE
INSUFFICIENT RELIABLE EVIDENCE to RATE
INSUFFICIENT RELIABLE EVIDENCE to RATE
Below is general information about the safety of the known ingredients contained in the product Probiotic with Digestive Enzymes. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
LIKELY SAFE ...when used orally and appropriately. Bifidobacterium lactis has been safely used alone or in combination with other probiotics in clinical trials lasting up to 12 weeks (92255,98502,105158,107572,107581,107586,110979,110985,110986,110992)(110993,110998,110999).
CHILDREN: LIKELY SAFE
when used orally and appropriately in children of most ages.
Bifidobacterium lactis has been safely used alone or in combination with other probiotics in infants and children for up to 15 months (3169,3458,92265,95381,95382,98736,105149,107582,107583,107585)(107587,107590,110984,110987,110988,110991,110994,110995). A combination probiotic containing B. lactis and Lactobacillus acidophilus (HOWARU Protect, Danisco) has been used safely for up to 6 months in children aged 3-5 years (16847). A specific combination of B. lactis, Bifidobacterium bifidum, and L. acidophilus (Complete Probiotic Platinum) has also been used safely for up to 18 months in children aged 4 months to 5 years (103436). In addition, in children ages 4-17 years, 1 billion CFUs of a 1:1:1 combination of B. lactis CECT 8145, Lacticasebacillus casei CECT 9104, and Bifidobacterium longum CECT 7347 has been used safely for 12 weeks (107531). There is insufficient reliable information available about the safety of B. lactis in preterm infants with a birth weight under 1000 grams. Cases of bacteremia have occurred rarely in preterm infants given other probiotics (102416,111610,111612,111613,111850,111852,111853). The US Food and Drug Administration (FDA) has issued a warning about cases of serious infections caused by probiotics reported in very preterm or very low birth weight infants under 1000 grams (111610). Similarly, the American Academy of Pediatrics does not support the routine administration of probiotics to these infants due to conflicting data on safety and efficacy (111608).
PREGNANCY AND LACTATION:
Insufficient reliable information available.
A meta-analysis of four clinical trials shows that taking probiotics during pregnancy increases the relative risk of pre-eclampsia by 85% when compared with placebo. Although the specific effects of Bifidobacterium lactis are unclear from this analysis, three of the included studies used B. lactis in combination with Lacticaseibacillus rhamnosus (105185). More information is needed to determine if certain patients are at increased risk.
LIKELY SAFE ...when used orally and appropriately. Bifidobacterium longum has been safely used alone or in combination with other probiotics in clinical trials lasting up to one year (1233,12108,13054,14334,35382,35403,35424,103440,103446,105129)(107593,110968,110972,111773,111776,111847,111851,111854,111857,111858).
CHILDREN: LIKELY SAFE
when used orally and appropriately in children of most ages.
Bifidobacterium longum has been safely used alone or in combination with other probiotics in infants and children for up to 4 months (3162,35377,35383,35393,35406,35407,92266,98736,107531,110924)(110976,111001,111015,111825,111833,111848). There is insufficient reliable information available about the safety of B. longum in preterm infants with a birth weight under 1000 grams. Cases of bacteremia have occurred rarely in preterm infants given these and other probiotics (102416,111610,111612,111613,111850,111852,111853). The US Food and Drug Administration (FDA) has issued a warning about cases of serious infections caused by probiotics reported in very preterm or very low birth weight infants under 1000 grams (111610). Similarly, the American Academy of Pediatrics does not support the routine administration of probiotics to these infants due to conflicting data on safety and efficacy (111608).
PREGNANCY AND LACTATION: POSSIBLY SAFE
when used orally and appropriately, short-term.
A combination of Bifidobacterium longum and Lacticaseibacillus rhamnosus has been used with apparent safety throughout pregnancy (105128,105144). A combination of B. longum BB536 and Bifidobacterium breve M-16V has been used with apparent safety from about 4 weeks before the expected due date until delivery (111015). Also, a combination of B. longum and Lacticaseibacillus paracasei has been used with apparent safety from 2 months prior to delivery until 2 months after delivery during lactation (90285).
LIKELY SAFE ...when used orally and appropriately. Lactobacillus acidophilus has been safely used as part of multi-ingredient probiotic products in studies lasting up to nine months (1731,6087,14370,14371,90231,90296,92255,103438,12775,107581)(110950,110970,110979,110998,111785,111793). ...when used intravaginally and appropriately. L. acidophilus has been used safely in studies lasting up to 12 weeks (12108,13176,13177,90265). There is insufficient reliable information available about the safety of non-viable, heat-killed L. acidophilus formulations when used orally.
CHILDREN: LIKELY SAFE
when used orally and appropriately in children of most ages.
Lactobacillus acidophilus has been safely used for up to 5 days (96887). Also, combination probiotics containing L. acidophilus have been used with apparent safety in various doses and durations. L. acidophilus has been combined with Bifidobacterium animalis (HOWARU Protect, Danisco) for up to 6 months in children 3-5 years old (16847), with Bifidobacterium bifidum for 6 weeks (90602,96890), with Bifidobacterium bifidum and Bifidobacterium animalis subsp. lactis (Complete Probiotic Platinum) for 18 months in children 4 months to 5 years of age (103436), and in a specific product (Visbiome, ExeGi Pharma) containing a total of 8 species for 3 months in children 2-12 years old (107497). There is insufficient reliable information available about the safety of L. acidophilus in preterm infants with a birth weight under 1000 grams. Cases of bacteremia have occurred rarely in preterm infants given other probiotics (102416,111610,111612,111613,111850,111852,111853). The US Food and Drug Administration (FDA) has issued a warning about cases of serious infections caused by probiotics reported in very preterm or very low birth weight infants under 1000 grams (111610). Similarly, the American Academy of Pediatrics does not support the routine administration of probiotics to these infants due to conflicting data on safety and efficacy (111608).
PREGNANCY: POSSIBLY SAFE
when used orally and appropriately.
A combination of Lactobacillus acidophilus, Lacticaseibacillus casei, and Bifidobacterium bifidum has been used with apparent safety for 6 weeks, starting at 24-28 weeks' gestation (95416,98430).
LACTATION:
There is insufficient reliable information available about the safety of Lactobacillus acidophilus during lactation.
However, there are currently no reasons to expect safety concerns when used appropriately.
LIKELY SAFE ...when prescription pancreatic enzyme products are used orally and appropriately under the guidance of a healthcare professional (98667,98674,98676,98677,99115,99116,99118,99120,99122,99124)(99125,99126,99127). Prescription pancreatic enzyme products are typically initiated at a dose of 500-1000 lipase units/kg body weight per meal to a maximum of 2500 lipase units/kg body weight per meal or 4000 lipase units/gram of fat daily. Doses higher than 2500 lipase units/kg body weight per meal are prescribed only if medically necessary (99130).
POSSIBLY UNSAFE ...when prescription pancreatic enzyme products are used orally at doses over 2500 lipase units/kg body weight per meal or 10,000 lipase units/kg body weight daily. Higher doses, especially those greater than 6000 lipase units/kg body weight per meal, have been associated with fibrosing colonopathy (99130). There is insufficient reliable information available about the safety of supplemental forms of pancreatic enzyme products.
CHILDREN: LIKELY SAFE
when prescription pancreatic enzyme products are used orally and appropriately under the guidance of a healthcare professional (99118,99119,99120,99121,99122,99124,99125,99126).
Prescription pancreatic enzyme products are usually initiated at a dose of 500-1000 lipase units/kg body weight per meal to a maximum of 2500 lipase units/kg body weight per meal or 4000 lipase units/gram fat daily. Doses higher than 2500 lipase units/kg body weight per meal are prescribed only if medically necessary (99130).
CHILDREN: POSSIBLY UNSAFE
when prescription pancreatic enzyme products are used orally at doses over 2500 lipase units/kg body weight per meal or 10,000 lipase units/kg body weight daily.
Higher doses, especially those greater than 6000 lipase units/kg body weight per meal, have been associated with fibrosing colonopathy (99130). There is insufficient reliable information available about the safety of supplemental forms of pancreatic enzyme products.
PREGNANCY AND LACTATION:
Insufficient reliable information available; avoid using unless essential for replacement therapy (15).
Below is general information about the interactions of the known ingredients contained in the product Probiotic with Digestive Enzymes. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
Theoretically, taking Bifidobacterium lactis with antibiotic drugs might decrease the effectiveness of B. lactis.
|
Theoretically, taking Bifidobacterium longum with antibiotic drugs might decrease the effectiveness of B. longum.
|
Theoretically, taking Lactobacillus acidophilus with antibiotic drugs might decrease the effectiveness of L. acidophilus.
L. acidophilus preparations usually contain live and active organisms. Therefore, simultaneously taking antibiotics might kill a significant number of the organisms (1740). Tell patients to separate administration of antibiotics and L. acidophilus preparations by at least two hours.
|
Theoretically, pancreatic enzyme products may reduce the effects of acarbose.
The digestive enzymes present in pancreatic enzyme products may break down acarbose, reducing its effects (9).
|
Below is general information about the adverse effects of the known ingredients contained in the product Probiotic with Digestive Enzymes. Some ingredients may not be listed. This information does NOT represent a recommendation for or a test of this specific product as a whole.
General
...Orally, Bifidobacterium lactis seems to be well tolerated by most patients.
Most Common Adverse Effects:
Orally: Diarrhea.
Serious Adverse Effects (Rare):
Orally: There is concern that probiotics may cause infections in some people.
Dermatologic ...In clinical research, two cases of rash, one with itching, were reported by patients taking a combination of Bifidobacterium lactis BB-12, Lacticaseibacillus paracasei F19, and Lactobacillus acidophilus La5. However, it is not clear if these adverse effects were due to B. lactis, other probiotics, or the combination, or if the events were idiosyncratic (90236).
Gastrointestinal ...Bloating and flatulence have been reported with probiotic use; however, these adverse effects have not been reported from ingestion of Bifidobacterium lactis in particular. When taken orally, B. lactis can cause diarrhea and other gastrointestinal complaints in children (3169,95381,105149). Gastrointestinal complaints including worsening diarrhea, abdominal pain, constipation, stomach burn, and flatulence have been reported rarely (110986,110999).
Immunologic
...There have been cases of Bifidobacterium bacteremia in critically ill patients (102416,107599).
These cases are rare and none seem to be due to Bifidobacterium lactis alone.
A specific preparation (NBL probiotic ATP, Nobel) containing B. lactis, Lacticaseibacillus casei, Lacticaseibacillus rhamnosus, Lactiplantibacillus plantarum, fructo-oligosaccharides, galacto-oligosaccharides, colostrum, and lactoferrin was found to be a significant risk factor for vancomycin-resistant Enterococcus colonization in premature infants. Although there was no direct link to determine causation, it was hypothesized that the probiotic mixture helped to mediate the acquisition and transfer of antibiotic resistance genes (96890).
General
...Orally, Bifidobacterium longum seems to be well tolerated by most patients.
Serious Adverse Effects (Rare):
Orally: There is concern that B. longum may cause bacteremia in certain patients.
Gastrointestinal ...When taken orally, abdominal discomfort, pain, and distension have been reported rarely (111773,111847,111856). Flatulence has been reported rarely with Bifidobacterium longum when used alone or in combination with other species of probiotics (107520,111773). Other rare gastrointestinal side effects have included constipation and gastrointestinal motor disorder (111773).
Immunologic ...There have been rare cases of Bifidobacterium bacteremia in critically ill infant and adult patients (102416,107599). Various cases of Bifidobacterium longum bacteremia, sometimes presenting as sepsis, have occurred in preterm infants using probiotics (102416,111610,111612,111850,111852,111853). In one case report, a 15-month-old female infant with congenital heart defects and recent surgery to replace a mechanical heart valve developed Bifidobacterium sepsis after being treated with IV antibiotics, extracorporeal membrane oxygenation (ECMO), and oral probiotics containing B. longum. It was thought that ECMO contributed to translocation of bifidobacteria from the gut and into the blood (102416). In 5 cases, very-low birthweight preterm infants developed B. longum bacteremia following the use of a specific probiotic product providing B. longum and Lactobacillus acidophilus (Infloran) for the prevention of necrotizing enterocolitis; antibiotic treatment was required in at least some of the cases (111850,111852,111853). Cases of sepsis related to B. longum have also occurred in adults; however, association with supplementation is unlikely. In one case, sepsis with B. longum occurred following acupuncture. This was likely due to needle contamination and not to supplementation (1236). In another case, a 71-year-old male with liver cirrhosis and prostate cancer developed B. longum lumbar vertebrodiscitis. The source was thought to be translocation from the intestine (111859). A 42-year-old male developed B. longum peritonitis secondary to intestinal perforation (111855).
Pulmonary/Respiratory ...When taken orally, a dry cough has been reported by a single patient in a clinical trial (111851).
Other ...When taken orally, weight gain has been reported by a single patient in a clinical trial (111773).
General
...Orally and intravaginally, Lactobacillus acidophilus is generally well tolerated.
Most Common Adverse Effects:
Orally: Mild gastrointestinal adverse effects.
Intravaginally: Vaginal discharge.
Serious Adverse Effects (Rare):
Orally: There is concern that L. acidophilus may cause infections in some people.
Dermatologic ...Orally, in one clinical trial, a combination of Lactobacillus acidophilus La-5, Lacticaseibacillus paracasei subsp. paracasei F19, and Bifidobacterium animalis subsp. lacltis BB-12 was associated with two cases of rash, one with itching. However, it is not clear if these adverse effects were due to L. acidophilus, other ingredients, the combination, or if the events were idiosyncratic (90236).
Gastrointestinal ...Orally, taking Lactobacillus acidophilus in combination with other probiotics may cause gastrointestinal side effects including epigastric discomfort (90239), abdominal pain (90239,90291,111785), dyspepsia (90239), flatulence (107497,107520), bloating (107497,111785), diarrhea (111785), vomiting (107537), and burping (90239); however, these events are uncommon.
Genitourinary ...Intravaginally, cream containing Lactobacillus acidophilus has been shown to cause increased vaginal discharge in about 5% of patients, compared to about 1% of patients receiving placebo cream (90237). Vaginal burning was reported by one person using intravaginal L. acidophilus and Limosilactobacillus fermentum in a clinical trial (111781).
Immunologic ...Since Lactobacillus acidophilus preparations contain live and active microorganisms, there is some concern that they might cause pathogenic infection in some patients. L. acidophilus has been isolated in some cases of bacteremia, sepsis, splenic abscess, liver abscess, endocarditis, necrotizing fasciitis, pancreatic necrosis, and meningoencephalitis. Most of these cases are thought to be due to the translocation of bacteria from other locations in the body in which they occur naturally, such as the oral cavity and gastrointestinal tract (107543,111782,111792). L. acidophilus endophthalmitis has been reported rarely (111787,111795). In one case, it was related to intravitreal injections for age-related macular degeneration in a 90-year-old female with an intraocular lens (111787). In another, it occurred following cataract surgery (111795).
General
...Orally, prescription pancreatic enzyme products are generally well tolerated when used at prescribed doses in adults and children.
Most Common Adverse Effects:
Orally: Prescription pancreatic enzyme products can cause abdominal pain, constipation, diarrhea, dry mouth, flatulence, irritation of the skin around the mouth and anus, nausea, steatorrhea, and vomiting.
Topically: Pancreatic enzyme product powder is irritating to the skin, eyes, mucus membranes, and respiratory tract. Pancreatic enzyme products that are held in the mouth prior to swallowing can cause irritation of the mucosa, including ulceration and stomatitis.
Serious Adverse Effects (Rare):
Orally: Prescription pancreatic enzyme products can worsen glucose control. Extremely high doses have been associated with fibrosing colonopathy and high uric acid levels in blood and urine.
Topically: Inhalation of dust containing pancreatic enzyme products has been associated with asthma, bronchospasm, and pulmonary hypersensitivity reactions.
Dermatologic
...Orally, rash or skin conditions have been reported rarely in individuals taking prescription pancreatic enzyme products (67709,98667).
A pruritic rash occurred in one patient taking a prescription pancreatic enzyme product in one clinical trial; this event was considered by the investigators to be possibly related to treatment (98667). Erythema also occurred in an 84-year-old patient who had taken a digestive enzyme supplement containing pancreatic enzymes and other enzymes (67709). In a case report, taking a supplemental form of pancreatic enzymes also containing hemicellulose and dried ox bile extract powder (Festal, Handok Inc) was thought to be the cause of an acute generalized exanthematous pustulosis (AGEP). The patient required an antihistamine and an oral and topical corticosteroid. Since this product contained more than one ingredient, the role of the pancreatic enzymes in this specific adverse event is unclear (98675).
Topically, exposure to pancreatic enzyme product powder is irritating to the skin. Hypersensitivity reactions such as skin rash have been reported (15).
Endocrine ...Orally, prescription pancreatic enzyme products may cause hypoglycemia or hyperglycemia in some patients (67622,98676,98677). In a double-blind trial of a prescription pancreatic enzyme product compared with placebo in patients with unresectable pancreatic cancer and associated pancreatic duct obstruction, one patient developed diabetes during the study period (67708).
Gastrointestinal
...Orally, prescription pancreatic enzyme products can cause abdominal pain, constipation, diarrhea, dry mouth, irritation of the skin around the mouth and anus, flatulence, nausea, steatorrhea, and vomiting.
However, these symptoms can be controlled by following prescription dosing recommendations (67617,67618,67645,67648,67651,67653,67657,67666,67714,98667)(98676,99115,99117,99123,99130). Doses of higher than 2500 lipase units/kg body weight per meal are not recommended unless medically necessary in order to prevent serious side effects, including fibrosing colonopathy and colonic strictures. These side effects are associated with the higher doses of lipase in prescription pancreatic enzyme products (2382,67677,67678,67679,67680,67682,67689,67690,67696,67701,99130).
Topically, pancreatic enzyme products that are held in the mouth prior to swallowing can cause irritation of the mucosa, including ulceration and stomatitis (15).
Hematologic ...Orally, severe neutropenia has been reported with a pancreatic enzyme product. In one case report, a 61-year-old patient taking a prescription pancreatic enzyme product for chronic pancreatitis developed agranulocytosis that persisted despite a trial of filgrastim. Upon discontinuation of the pancreatic enzyme product, marked improvement in neutropenia occurred within 7 days, with full resolution within 6 weeks (107411).
Neurologic/CNS ...Orally, headache and dizziness have been reported rarely in individuals taking prescription pancreatic enzyme products (67618,98667,99120).
Ocular/Otic ...Topically, exposure to pancreatic enzyme product powder is irritating to the eyes. Hypersensitivity reactions such as watery eyes have been reported (15).
Pulmonary/Respiratory ...Topically, exposure to pancreatic enzyme product powder is irritating to the respiratory tract. Hypersensitivity reactions such as sneezing have been reported (15). Inhalation of dust containing pancreatic enzyme products has been associated with allergic rhinitis, asthma, bronchospasm, and pulmonary hypersensitivity reactions (11768,11769,67641).
Renal ...Orally, extremely high doses of pancreatic enzyme products containing more than 10,000 lipase units/kg body weight daily have been associated with high uric acid levels in blood and urine (67673,67693,67705). Rarely, kidney pain has been reported (98667).
